Tissue/Cell/Blood Genomic DNA Extraction Kit KB1100
KB1100
Suitable for tissue, cell, blood DNA extraction
Product Information
Product Name | Cat. No. | Spec. |
Tissue/Cell/Blood Genomic DNA Extraction Kit | KB1100-50T | 50 T |
Product Description/Introduction
This kit releases genomic DNA from tissue cells through a specially optimized lysis buffer, and then purifies genomic DNA through a specific DNA-binding centrifugal column. It is suitable for isolating genomic DNA from animal tissues, cultured cells and whole blood. 10-30 μg high purity and complete genomic DNA can be rapidly extracted and purified from 2-30 mg animal tissues, 106-107 freshly cultured cell suspensions and 5-100 μL whole blood of non-nucleated or nucleated red cells (including anticoagulant). The obtained genomic DNA can be directly used for subsequent Molecular biology experiments such as PCR templates, digestion and hybridization.
Storage and Shipping Conditions
Proteinase K and RNase A should be shipped with wet ice, and stored at -20℃; Other reagents are shipped and stored at room temperature for up to 12 months.
Product Contents
Component Number | Component | KB1100-50T |
KB1100-1 | Buffer GA | 12 mL |
KB1100-2 | Proteinase K | 1 mL |
KB1100-3 | RNase A | 200 uL |
KB1100-4 | Buffer GB | 12 mL |
KB1100-5 | Buffer PD | 12 mL (18 mL of anhydrous ethanol added before use) |
KB1100-6 | Buffer PW | 24 mL (56 mL of anhydrous ethanol added before use) |
KB1100-7 | Buffer TE | 10 mL |
KB3633-8 | DNA Spin Columns(with Collection Tubes) | 50 |
Manual | One copy | |
Before starting (please read carefully)
1. If a precipitate has formed in Buffer GA, heat it at 65°C until the precipitate has fully dissolved and use it after it returns to room temperature..
2. Before first use, add 18 mL of absolute ethanol to Buffer PD, add 56 mL of absolute ethanol to Buffer PW and mix well separately.
Assay Protocol / Procedures
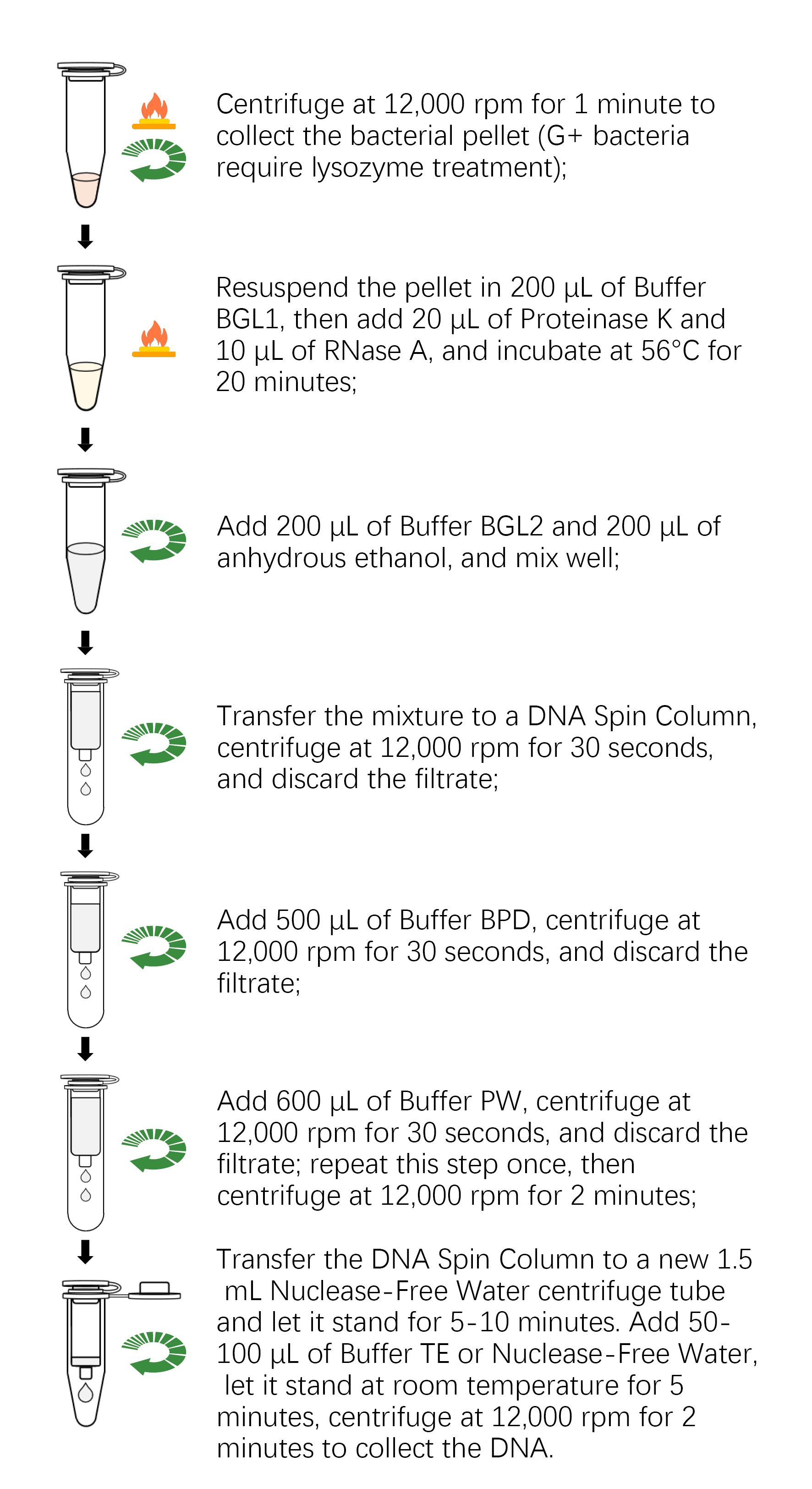
1. Samples Preparation
a. Take out 2-30 mg of fresh or frozen animal tissue, place them in a 1.5 mL centrifuge tube or special grinding tube containing 2-3 grinding beads with a diameter of 3 mm, add 200 μL of Buffer GA, and use a Tissue Homogenizer to thoroughly grind the tissue to homogenization at room temperature (if the tissue is not thoroughly homogenized, it will affect the yield and quality of DNA).
b. Take 10 6-10 7 of cell suspension into a 1.5 mL centrifuge tube, centrifuge at 1,000 rpm for 5 minutes, gently aspirate and discard supernatant with a pipette. Add 200 μL of Buffer GA, mix well by votex.
c. Take 50-100 μL whole blood of non-nucleated erythrocytes (including anticoagulant) into a 1.5 mL centrifuge tube, add Buffer GA solution to 200 μL, mix well with votex.
Note: For processing a larger volume of blood, add 3 times volume of erythrocyte lysate to the sample and mix it upside down, let it stand at room temperature for 5 min, mix it upside down several times during incubation, Centrifuge it at 3,000 rpm for 10 min, gently remove the supernatant with pipette to leave the white blood cell precipitate, add 200 μL Buffer GA, mix well with votex.
d. Take 5-20 μL whole blood of nucleated erythrocytes (including anticoagulant) into a 1.5 mL centrifuge tube, add Buffer GA solution to 200 μL, mix well with votex.
2. Add 20 μL of Proteinase K and 4 μL of RNase A, incubate at 56℃ for 30 minutes (Mix every 10 minutes to accelerates tissue lysis).
Note: Please extend time for materials that are difficult to lysis.
3. Centrifuge at 12,000 rpm for 2 min, transfer the tissue supernatant to a Nuclease-free centrifuge tube, avoiding aspirating the precipitate. (Please skip this step if the sample is cell or blood).
4. Add 200 μL of Buffer GB to the centrifuge tube, mix upside and down, and incubate at 70 °C for 10 min.
5. Add 200 μL of absolute ethanol to the centrifuge tube, mix upside and down (any visible precipitate that may form), and then centrifuge at 12,000 rpm for 2 min at room temperature.
6. Transfer the above supernatant into the DNA Spin Columns.
7. Centrifuge at 12,000 rpm for 30 seconds, discard the flow-through and put the DNA Spin Columns back into the Collection Tube.
8. Add 500 μL Buffer PD to the DNA Spin Columns, centrifuge at 12,000 rpm for 30 seconds, discard the flow-through.
9. Add 600 μL Buffer PW to the DNA Spin Columns (please add Buffer PW along the wall of the tube to help rinse out any residual salts on the tube wall), centrifuge at 12,000 rpm for 30 seconds, discard the flow-through.
10. Repeat Step 9.
11. Put the DNA Spin Column in a Collection Tube and centrifuge at 12,000 rpm for 2 minutes.
12. Put the DNA Spin Columns into a new Nuclease-free 1.5 mL centrifuge tube, stand at room temperature for 5-10 minites to allow the ethanol residue from the DNA Spin Column to completely evaporate.
13. Add 50-100 μL Buffer TE or Nuclease-free Water to the center of the membrane of the DNA Spin Column and leave it at room temperature for 5 min (Pre-heating the Buffer TE or Nuclease-free Water at 65℃ can improve elution efficiency).
14. Centrifuge at 12,000 rpm for 2 minutes to collect DNA. To obtain a higher concentration of DNA, the first flow-through can be re-added into the DNA Spin Column and let it stand for 5 minutes at room temperature and centrifuge at 12,000 rpm for 2 minutes, and the DNA collected again..
Note:
1. Please read the Product Manual carefully before use.
2. Fresh materials should be prepared to ensure that the genomic DNA is not degraded.
3. Repeated freezing and thawing of samples should be avoided, as this may result in a decrease in the yield of DNA.
4. Tissue material should never exceed the maximum starting amount and should be sufficiently lysed, otherwise it may affect the yield or even clog the column.
For Research Use Only!
Operation Flow





 sales
sales